
It is a rectangular grid of the elements, with periods going across and families going down. 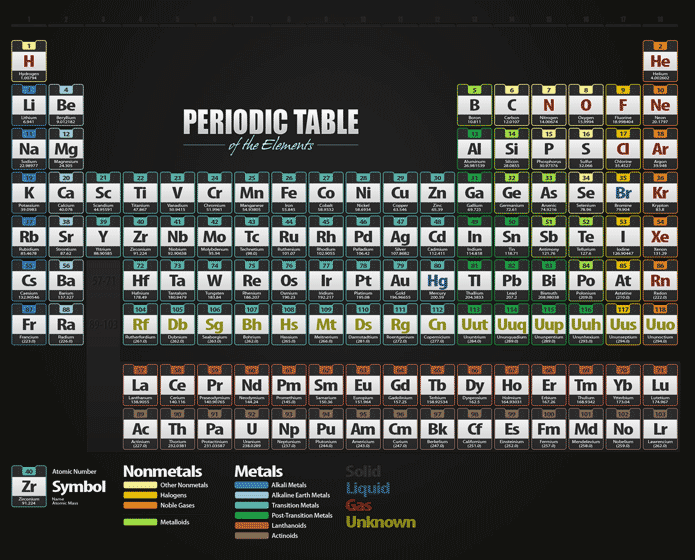
The form of the table that is most familiar to us today is usually referred to as the short-form periodic table, or the common (or standard) form of the table. Not only did he come up with the organization of them, but he also predicted the existence of many elements before they were discovered. He had been experimenting with organizing the elements according to their chemical properties and eventually noticed that certain trends existed within groups of elements. The periodic table was first published by Dimitri Mendeleev in 1869. And each element is represented by a symbol which is usually one or two letters taken from the name. All the elements of these groups have their own properties and reactivities.Ĭolor coding can be used to distinguish between columns (groups). The groups between these two columns are alkaline-earth metals, transition metal groups, inner transition metal groups, non-metal groups, halogens. At the left of the table is a group that contains reactive metals, such as sodium and potassium. Each row of elements in the periodic table is called a period, and the elements are grouped according to the fill of electrons in their valence shells.Īt the right of the table is a group that contains inert gases elements with full valence shells and which are generally unreactive. It is also ordered by element block according to electron shell configuration. 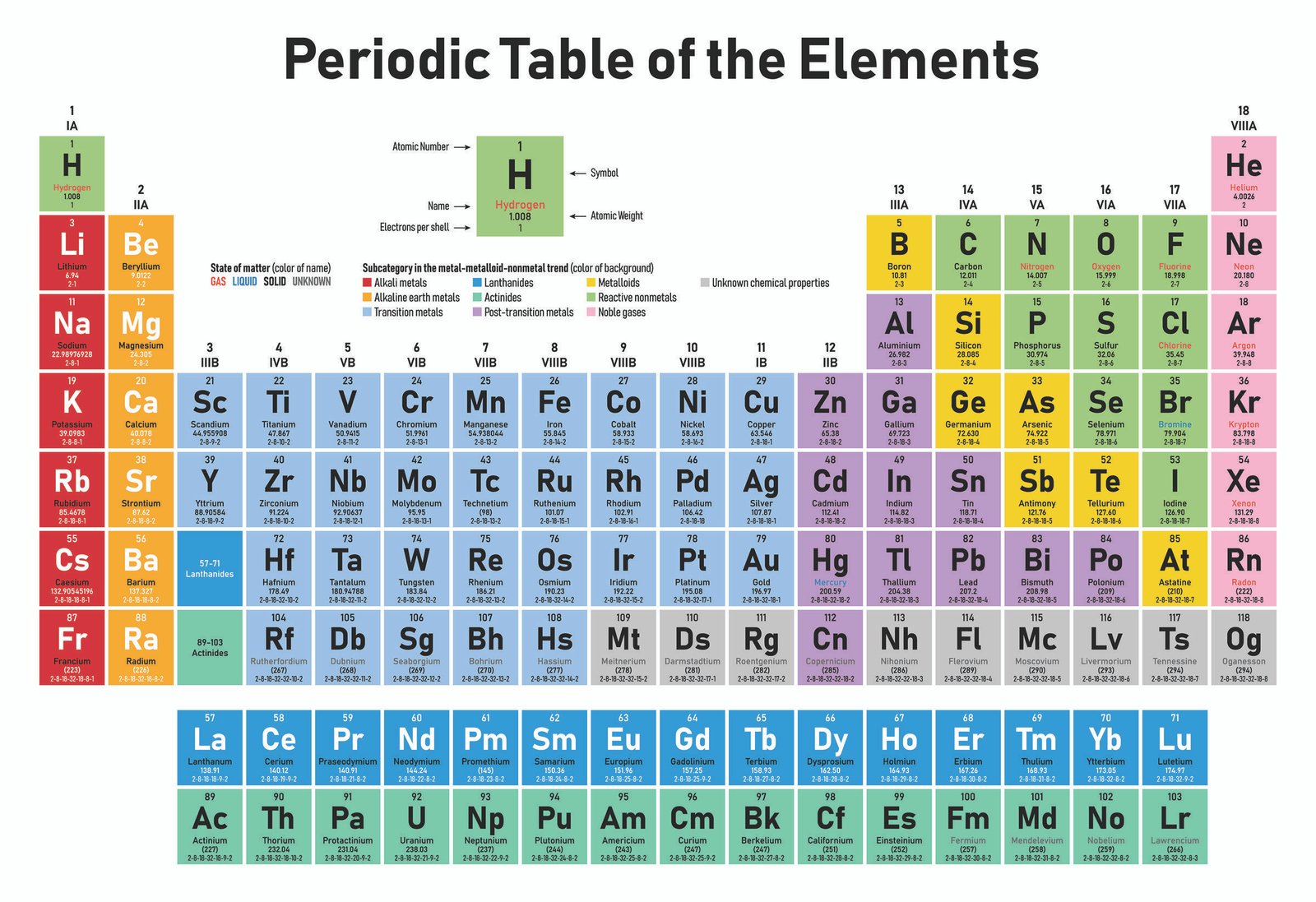 
It is organized by atomic number with hydrogen being number 1, helium being number 2, etc. These two categories go from left to right across horizontal rows (known as a period), and up & down within vertical columns (known as a group). The periodic table is called the "Periodic Table" because it's organized into periods and groups. The periodic table can most simply be described as a chart that organizes the chemical elements of matter into an easy-to-identify visual flow chart. Elements that exhibit similar chemistry appear in vertical columns called groups (numbered 1–18 from left to right) the seven horizontal rows are called periods.What is a Simple Definition of the Periodic Table? It arranges of the elements in order of increasing atomic number. The periodic table is used as a predictive tool. As expected, semimetals exhibit properties intermediate between metals and nonmetals. Most solid nonmetals are brittle, so they break into small pieces when hit with a hammer or pulled into a wire. Nonmetals can be gases (such as chlorine), liquids (such as bromine), or solids (such as iodine) at room temperature and pressure. Nonmetals, in contrast, are generally poor conductors of heat and electricity and are not lustrous. Of the metals, only mercury is a liquid at room temperature and pressure all the rest are solids. The vast majority of the known elements are metals. Metals-such as copper or gold-are good conductors of electricity and heat they can be pulled into wires because they are ductile they can be hammered or pressed into thin sheets or foils because they are malleable and most have a shiny appearance, so they are lustrous. The distinction between metals and nonmetals is one of the most fundamental in chemistry. Gold-colored lements that lie along the diagonal line exhibit properties intermediate between metals and nonmetals they are called semimetals. \) divides the elements into metals (in blue, below and to the left of the line) and nonmetals (in bronze, above and to the right of the line).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
